Luxol Fast Blue staining¶
Contributed by Luke Hammond, QBI, The University of Queensland, Australia
Luxol Fast Blue histological stain. This technique is a method requiring over-staining of tissues and differentiation to reveal the myelin. It is advisable to include a control showing de-myelination pathology to ensure the end point of staining is reached. Cresyl Fast Violet counter-stain is optional and results in neuronal piliform staining and a deepening of the myelin blue staining.
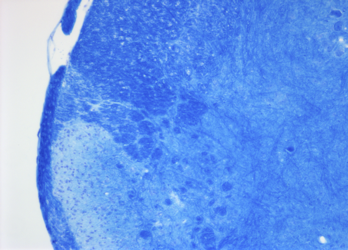
Cresyl Fast Violet counter-stain is optional and results in neuronal piliform staining and a deepening of the myelin blue staining.
Requirements¶
Mounted sections on glass slides Slide staining rack, Staining jars, Coplin jar Absolute alcohol Xylene Distilled water. DePeX Luxol Fast Blue (Fronine: FG080) Cresyl Fast Violet 0.2% (Fronine: HH155) Lithium Carbonate (Sigma Aldrich: 62470) 0.05% Lithium Carbonate(aq)
Method¶
- Heat the staining solution to 60°C in the incubator (this will take 30mins approximately depending on the volume to be heated.
- Dewax or defat sections prior to rehydration as necessary. Rinse in tap water.
- Immerse the slides in the LFB solution and incubate at 60°C for 4 hours.
- Rinse off excess stain in distilled water
- Differentiate slides in Lithium Carbonate for 30 seconds
- Rinse in distilled water.
- Check staining microscopically and repeat steps 4 to 5 until myelinated and unmyelinated regions are defined.
- Counterstain with Cresyl Fast Violet for 8mins
- Dehydrate through 95% ethanol and check the Cresyl Fast Violet staining at that point. (70% ethanol will continue to remove the LFB).
- Dehydrate through absolute ethanol, clear in xylene and mount in DPX
This method is based, with permission, on an original protocol available here.